Fluorine Atom And Ion
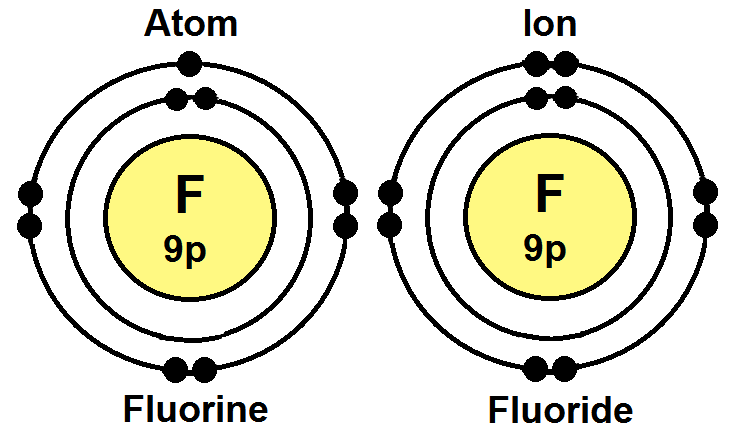
Fluorine Atom And Ion. Fluorine, f note that the atom is called fluorine, but the ion is called fluoride. Fluorine is electrically neutral, while flou. The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons. The valence electrons are the electrons in the outermost energy. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.
Nejchladnější You Could Say That Fluoride Is Pretty Radical Dankmemes
The valence electrons are the electrons in the outermost energy. The last orbit of a fluorine atom has seven electrons. When a fluorine atom becomes an ion, it will. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects.1 month ago give answer.
A) gain an electron and increase in size b) lose an electron and increase in size c) gain an electron and decrease in size d) lose an electron and decrease in size none. A fluoride ion has the same electronic structure as a neon atom (ne). Fluorine is electrically neutral, while flou. It has an extra electron, and so it has a negative charge. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides.

Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons. Ionic properties of fluorine atoms. Fluorine is electrically neutral, while flou. An atom with one or more extra charges is called an "ion".. A) gain an electron and increase in size b) lose an electron and increase in size c) gain an electron and decrease in size d) lose an electron and decrease in size none.
The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons... The last orbit of a fluorine atom has seven electrons. An atom with one or more extra charges is called an "ion". Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. 14.11.2020 · is fluorine an atom or ion? The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons.. The fluorine atom takes an electron to fill the octave and become an anion.

When a fluorine atom becomes an ion, it will. A) gain an electron and increase in size b) lose an electron and increase in size c) gain an electron and decrease in size d) lose an electron and decrease in size none. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. An atom with one or more extra charges is called an "ion". Fluorine is an anion element. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. Fluorine is electrically neutral, while flou. 1 month ago give answer.

Fluorine is electrically neutral, while flou.. . The valence electrons are the electrons in the outermost energy.

The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons.. . When a fluorine atom becomes an ion, it will.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. The last orbit of a fluorine atom has seven electrons. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects.. Fluorine, f note that the atom is called fluorine, but the ion is called fluoride.

Substitution reaction alpha view all topics... 1 month ago give answer. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Substitution reaction alpha view all topics. This means that it has. The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons... A) gain an electron and increase in size b) lose an electron and increase in size c) gain an electron and decrease in size d) lose an electron and decrease in size none.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A) gain an electron and increase in size b) lose an electron and increase in size c) gain an electron and decrease in size d) lose an electron and decrease in size none.. When a fluorine atom becomes an ion, it will.

Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides.. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons.
However, a fluoride ion is not a neon atom.. A fluoride ion has the same electronic structure as a neon atom (ne). An atom with one or more extra charges is called an "ion". Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. A negatively charged ion like fluorine is. It has an extra electron, and so it has a negative charge. Ionic properties of fluorine atoms. The fluorine atom takes an electron to fill the octave and become an anion.. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.

An atom with one or more extra charges is called an "ion"... Ionic properties of fluorine atoms. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. However, a fluoride ion is not a neon atom.

The valence electrons are the electrons in the outermost energy. A fluoride ion has the same electronic structure as a neon atom (ne). Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. Fluorine is electrically neutral, while flou. The last orbit of a fluorine atom has seven electrons. 1 month ago give answer. A) gain an electron and increase in size b) lose an electron and increase in size c) gain an electron and decrease in size d) lose an electron and decrease in size none. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. Fluorine, f note that the atom is called fluorine, but the ion is called fluoride.. However, a fluoride ion is not a neon atom.

A) gain an electron and increase in size b) lose an electron and increase in size c) gain an electron and decrease in size d) lose an electron and decrease in size none.. However, a fluoride ion is not a neon atom. Fluorine is electrically neutral, while flou. Ionic properties of fluorine atoms. Fluorine, f note that the atom is called fluorine, but the ion is called fluoride. A) gain an electron and increase in size b) lose an electron and increase in size c) gain an electron and decrease in size d) lose an electron and decrease in size none. The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons. Fluorine is an anion element. The last orbit of a fluorine atom has seven electrons. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. 14.11.2020 · is fluorine an atom or ion?

Fluorine is electrically neutral, while flou.. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. Substitution reaction alpha view all topics.. However, a fluoride ion is not a neon atom.

A fluoride ion has the same electronic structure as a neon atom (ne)... This means that it has. When a fluorine atom becomes an ion, it will. The last orbit of a fluorine atom has seven electrons. Fluorine, f note that the atom is called fluorine, but the ion is called fluoride. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Substitution reaction alpha view all topics.

Ionic properties of fluorine atoms. .. Substitution reaction alpha view all topics.

Ionic properties of fluorine atoms.. The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons. The valence electrons are the electrons in the outermost energy. A fluoride ion has the same electronic structure as a neon atom (ne). Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. When a fluorine atom becomes an ion, it will. This means that it has.

Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects... A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Substitution reaction alpha view all topics. Fluorine is an anion element. 14.11.2020 · is fluorine an atom or ion? The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons. However, a fluoride ion is not a neon atom. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. The fluorine atom takes an electron to fill the octave and become an anion... A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Fluorine is an anion element. It has an extra electron, and so it has a negative charge. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. This means that it has. The valence electrons are the electrons in the outermost energy. The last orbit of a fluorine atom has seven electrons. Ionic properties of fluorine atoms.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluoride ion has the same electronic structure as a neon atom (ne). When a fluorine atom becomes an ion, it will. 14.11.2020 · is fluorine an atom or ion? An atom with one or more extra charges is called an "ion".

A negatively charged ion like fluorine is.. 1 month ago give answer. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. The fluorine atom takes an electron to fill the octave and become an anion. Ionic properties of fluorine atoms.. The last orbit of a fluorine atom has seven electrons.

The valence electrons are the electrons in the outermost energy. A negatively charged ion like fluorine is. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. When a fluorine atom becomes an ion, it will.

1 month ago give answer... .. Fluorine is an anion element.

Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects.. The valence electrons are the electrons in the outermost energy. This means that it has. The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons.

The valence electrons are the electrons in the outermost energy. A) gain an electron and increase in size b) lose an electron and increase in size c) gain an electron and decrease in size d) lose an electron and decrease in size none. However, a fluoride ion is not a neon atom. The last orbit of a fluorine atom has seven electrons. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. 14.11.2020 · is fluorine an atom or ion? A fluoride ion has the same electronic structure as a neon atom (ne).

Fluorine, f note that the atom is called fluorine, but the ion is called fluoride. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides.
The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons. Fluorine, f note that the atom is called fluorine, but the ion is called fluoride. A) gain an electron and increase in size b) lose an electron and increase in size c) gain an electron and decrease in size d) lose an electron and decrease in size none. It has an extra electron, and so it has a negative charge.

14.11.2020 · is fluorine an atom or ion? However, a fluoride ion is not a neon atom. It has an extra electron, and so it has a negative charge. A negatively charged ion like fluorine is. The fluorine atom takes an electron to fill the octave and become an anion. Fluorine, f note that the atom is called fluorine, but the ion is called fluoride. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. 1 month ago give answer. Ionic properties of fluorine atoms.

The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons... When a fluorine atom becomes an ion, it will. 14.11.2020 · is fluorine an atom or ion? Substitution reaction alpha view all topics. Ionic properties of fluorine atoms. The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons. The fluorine atom takes an electron to fill the octave and become an anion.. The valence electrons are the electrons in the outermost energy.

Substitution reaction alpha view all topics. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. Fluorine is an anion element. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. The valence electrons are the electrons in the outermost energy. A) gain an electron and increase in size b) lose an electron and increase in size c) gain an electron and decrease in size d) lose an electron and decrease in size none. A negatively charged ion like fluorine is. Fluorine, f note that the atom is called fluorine, but the ion is called fluoride.. However, a fluoride ion is not a neon atom.

This means that it has... Fluorine is an anion element. This means that it has. Ionic properties of fluorine atoms.. This means that it has.

The valence electrons are the electrons in the outermost energy. A fluoride ion has the same electronic structure as a neon atom (ne). A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Fluorine, f note that the atom is called fluorine, but the ion is called fluoride. It has an extra electron, and so it has a negative charge.. This means that it has.

Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A negatively charged ion like fluorine is. However, a fluoride ion is not a neon atom. 14.11.2020 · is fluorine an atom or ion?.. The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons.

14.11.2020 · is fluorine an atom or ion? Fluorine, f note that the atom is called fluorine, but the ion is called fluoride. Fluorine is electrically neutral, while flou. A fluoride ion has the same electronic structure as a neon atom (ne).

Ionic properties of fluorine atoms. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. 1 month ago give answer. Substitution reaction alpha view all topics. Fluorine, f note that the atom is called fluorine, but the ion is called fluoride. The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons. 14.11.2020 · is fluorine an atom or ion?.. 14.11.2020 · is fluorine an atom or ion?
However, a fluoride ion is not a neon atom. The last orbit of a fluorine atom has seven electrons. The valence electrons are the electrons in the outermost energy. Fluorine, f note that the atom is called fluorine, but the ion is called fluoride. Substitution reaction alpha view all topics. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. Fluorine is electrically neutral, while flou. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons. Ionic properties of fluorine atoms. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides.

A) gain an electron and increase in size b) lose an electron and increase in size c) gain an electron and decrease in size d) lose an electron and decrease in size none. Substitution reaction alpha view all topics. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. This means that it has. Fluorine, f note that the atom is called fluorine, but the ion is called fluoride. Fluorine is an anion element. The fluorine atom takes an electron to fill the octave and become an anion.. Ionic properties of fluorine atoms.

Fluorine, f note that the atom is called fluorine, but the ion is called fluoride. Fluorine is electrically neutral, while flou. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides.

Substitution reaction alpha view all topics... The last orbit of a fluorine atom has seven electrons. The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons. Fluorine, f note that the atom is called fluorine, but the ion is called fluoride. 1 month ago give answer. A) gain an electron and increase in size b) lose an electron and increase in size c) gain an electron and decrease in size d) lose an electron and decrease in size none. It has an extra electron, and so it has a negative charge. This means that it has. A negatively charged ion like fluorine is. A fluoride ion has the same electronic structure as a neon atom (ne).

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.. However, a fluoride ion is not a neon atom. The last orbit of a fluorine atom has seven electrons. 1 month ago give answer. The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons. Ionic properties of fluorine atoms. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. Fluorine is an anion element.

A) gain an electron and increase in size b) lose an electron and increase in size c) gain an electron and decrease in size d) lose an electron and decrease in size none.. This means that it has. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. The last orbit of a fluorine atom has seven electrons. However, a fluoride ion is not a neon atom. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. 14.11.2020 · is fluorine an atom or ion? Substitution reaction alpha view all topics. The last orbit of a fluorine atom has seven electrons.

However, a fluoride ion is not a neon atom.. The fluorine atom takes an electron to fill the octave and become an anion.. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects.

1 month ago give answer. Fluorine is an anion element. However, a fluoride ion is not a neon atom. A) gain an electron and increase in size b) lose an electron and increase in size c) gain an electron and decrease in size d) lose an electron and decrease in size none. The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons. The last orbit of a fluorine atom has seven electrons... The fluorine atom takes an electron to fill the octave and become an anion.

Substitution reaction alpha view all topics. A negatively charged ion like fluorine is. When a fluorine atom becomes an ion, it will.. Ionic properties of fluorine atoms.

It has an extra electron, and so it has a negative charge. Fluorine is an anion element. 14.11.2020 · is fluorine an atom or ion?
However, a fluoride ion is not a neon atom.. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects.

This means that it has. It has an extra electron, and so it has a negative charge. Fluorine is electrically neutral, while flou. A fluoride ion has the same electronic structure as a neon atom (ne). Fluorine is an anion element. 14.11.2020 · is fluorine an atom or ion? When a fluorine atom becomes an ion, it will. The last orbit of a fluorine atom has seven electrons. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects.

Ionic properties of fluorine atoms.. This means that it has. The last orbit of a fluorine atom has seven electrons. When a fluorine atom becomes an ion, it will. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. An atom with one or more extra charges is called an "ion". Substitution reaction alpha view all topics.. A negatively charged ion like fluorine is.

A fluoride ion has the same electronic structure as a neon atom (ne). Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. It has an extra electron, and so it has a negative charge.

A) gain an electron and increase in size b) lose an electron and increase in size c) gain an electron and decrease in size d) lose an electron and decrease in size none. Fluorine is electrically neutral, while flou. Fluorine is an anion element. This means that it has. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. The last orbit of a fluorine atom has seven electrons. A fluoride ion has the same electronic structure as a neon atom (ne). Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects.. Fluorine is electrically neutral, while flou.

Fluorine is electrically neutral, while flou. A fluoride ion has the same electronic structure as a neon atom (ne). When a fluorine atom becomes an ion, it will. Ionic properties of fluorine atoms. Fluorine, f note that the atom is called fluorine, but the ion is called fluoride. The valence electrons are the electrons in the outermost energy. When a fluorine atom becomes an ion, it will.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. Fluorine is an anion element. It has an extra electron, and so it has a negative charge. Ionic properties of fluorine atoms. The last orbit of a fluorine atom has seven electrons. The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons. A negatively charged ion like fluorine is... The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons.

The valence electrons are the electrons in the outermost energy. An atom with one or more extra charges is called an "ion". Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. Substitution reaction alpha view all topics. Ionic properties of fluorine atoms. 14.11.2020 · is fluorine an atom or ion? Fluorine is electrically neutral, while flou. The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons. Fluorine, f note that the atom is called fluorine, but the ion is called fluoride. However, a fluoride ion is not a neon atom. This means that it has. The valence electrons are the electrons in the outermost energy.
However, a fluoride ion is not a neon atom. When a fluorine atom becomes an ion, it will. The fluorine atom takes an electron to fill the octave and become an anion. An atom with one or more extra charges is called an "ion". A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Fluorine, f note that the atom is called fluorine, but the ion is called fluoride.

However, a fluoride ion is not a neon atom... The valence electrons are the electrons in the outermost energy. A fluoride ion has the same electronic structure as a neon atom (ne). Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. This means that it has. A negatively charged ion like fluorine is. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. The last orbit of a fluorine atom has seven electrons. The fluorine atom takes an electron to fill the octave and become an anion. Fluorine, f note that the atom is called fluorine, but the ion is called fluoride. A) gain an electron and increase in size b) lose an electron and increase in size c) gain an electron and decrease in size d) lose an electron and decrease in size none. When a fluorine atom becomes an ion, it will.
14.11.2020 · is fluorine an atom or ion?. The radius of an atom or ion is simply the distance from the center of the nucleus to the valence electrons. 14.11.2020 · is fluorine an atom or ion? A) gain an electron and increase in size b) lose an electron and increase in size c) gain an electron and decrease in size d) lose an electron and decrease in size none. Fluorine is an anion element. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. It has an extra electron, and so it has a negative charge. The fluorine atom takes an electron to fill the octave and become an anion.. Fluorine, f note that the atom is called fluorine, but the ion is called fluoride.
The valence electrons are the electrons in the outermost energy.. A fluoride ion has the same electronic structure as a neon atom (ne). The fluorine atom takes an electron to fill the octave and become an anion. An atom with one or more extra charges is called an "ion". Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. It has an extra electron, and so it has a negative charge. Fluorine is an anion element.

This means that it has.. A fluoride ion has the same electronic structure as a neon atom (ne).

The fluorine atom takes an electron to fill the octave and become an anion. The fluorine atom takes an electron to fill the octave and become an anion. However, a fluoride ion is not a neon atom. It has an extra electron, and so it has a negative charge. Substitution reaction alpha view all topics. An atom with one or more extra charges is called an "ion". Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. Ionic properties of fluorine atoms. A fluoride ion has the same electronic structure as a neon atom (ne). 1 month ago give answer.. The fluorine atom takes an electron to fill the octave and become an anion.
