Kolekce Neutral Nitrogen Atom
Kolekce Neutral Nitrogen Atom. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. There are 118 elements in the periodic table.
Prezentováno Solved Q 17 77 Which Of The Following Contain S Basic Chegg Com
24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge.
This element is found in group 15 and period 2 of the periodic table of the elements. This is the number of protons in the nuclei of nitrogen atoms. A neutral atom has the same number of electrons as protons. There are 118 elements in the periodic table. 20.06.2016 · the atomic number of nitrogen is 7. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.

This element is found in group 15 and period 2 of the periodic table of the elements. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … The lone pair of pyrrole is in such an. 20.06.2016 · the atomic number of nitrogen is 7. There are 118 elements in the periodic table.

24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7... For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … A neutral atom has the same number of electrons as protons. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. There are 118 elements in the periodic table. The lone pair of pyrrole is in such an. There are 118 elements in the periodic table. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons... This is the number of protons in the nuclei of nitrogen atoms.

For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. The lone pair of pyrrole is in such an. There are 118 elements in the periodic table. This is the number of protons in the nuclei of nitrogen atoms. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. It has an atomic … For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. A neutral atom has the same number of electrons as protons. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … There are 118 elements in the periodic table. A neutral atom has the same number of electrons as protons.

This is the number of protons in the nuclei of nitrogen atoms... 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. There are 118 elements in the periodic table. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. A neutral atom has the same number of electrons as protons. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he... For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …

Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge... For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. This element is found in group 15 and period 2 of the periodic table of the elements. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. The lone pair of pyrrole is in such an. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. It has an atomic …

This element is found in group 15 and period 2 of the periodic table of the elements. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring.
24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7... For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … There are 118 elements in the periodic table. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. 20.06.2016 · the atomic number of nitrogen is 7. A neutral atom has the same number of electrons as protons. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. This element is found in group 15 and period 2 of the periodic table of the elements. This is the number of protons in the nuclei of nitrogen atoms. 20.06.2016 · the atomic number of nitrogen is 7.

A neutral atom has the same number of electrons as protons. .. It has an atomic …

A neutral atom has the same number of electrons as protons... A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring.

It has an atomic … The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.

A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge... Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. A neutral atom has the same number of electrons as protons. The lone pair of pyrrole is in such an. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7.

This element is found in group 15 and period 2 of the periodic table of the elements. This element is found in group 15 and period 2 of the periodic table of the elements.

It has an atomic ….. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is ….. This element is found in group 15 and period 2 of the periodic table of the elements.

This element is found in group 15 and period 2 of the periodic table of the elements. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7.

The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. There are 118 elements in the periodic table. It has an atomic …. A neutral atom has the same number of electrons as protons.

Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge... For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.

There are 118 elements in the periodic table. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. This element is found in group 15 and period 2 of the periodic table of the elements. This is the number of protons in the nuclei of nitrogen atoms. A neutral atom has the same number of electrons as protons. It has an atomic … There are 118 elements in the periodic table. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3... It has an atomic …

Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge... The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. This is the number of protons in the nuclei of nitrogen atoms. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The lone pair of pyrrole is in such an. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. There are 118 elements in the periodic table. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge.

The lone pair of pyrrole is in such an. .. The lone pair of pyrrole is in such an.

Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. There are 118 elements in the periodic table.

For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring.. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. A neutral atom has the same number of electrons as protons. It has an atomic … 20.06.2016 · the atomic number of nitrogen is 7. This element is found in group 15 and period 2 of the periodic table of the elements. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … This is the number of protons in the nuclei of nitrogen atoms. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3... Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge.

This is the number of protons in the nuclei of nitrogen atoms.. 20.06.2016 · the atomic number of nitrogen is 7. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. The lone pair of pyrrole is in such an. This element is found in group 15 and period 2 of the periodic table of the elements. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. There are 118 elements in the periodic table.. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …

It has an atomic … 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. This is the number of protons in the nuclei of nitrogen atoms. A neutral atom has the same number of electrons as protons. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. 20.06.2016 · the atomic number of nitrogen is 7. There are 118 elements in the periodic table. This element is found in group 15 and period 2 of the periodic table of the elements. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7.

There are 118 elements in the periodic table... . For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …

The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. .. This element is found in group 15 and period 2 of the periodic table of the elements.
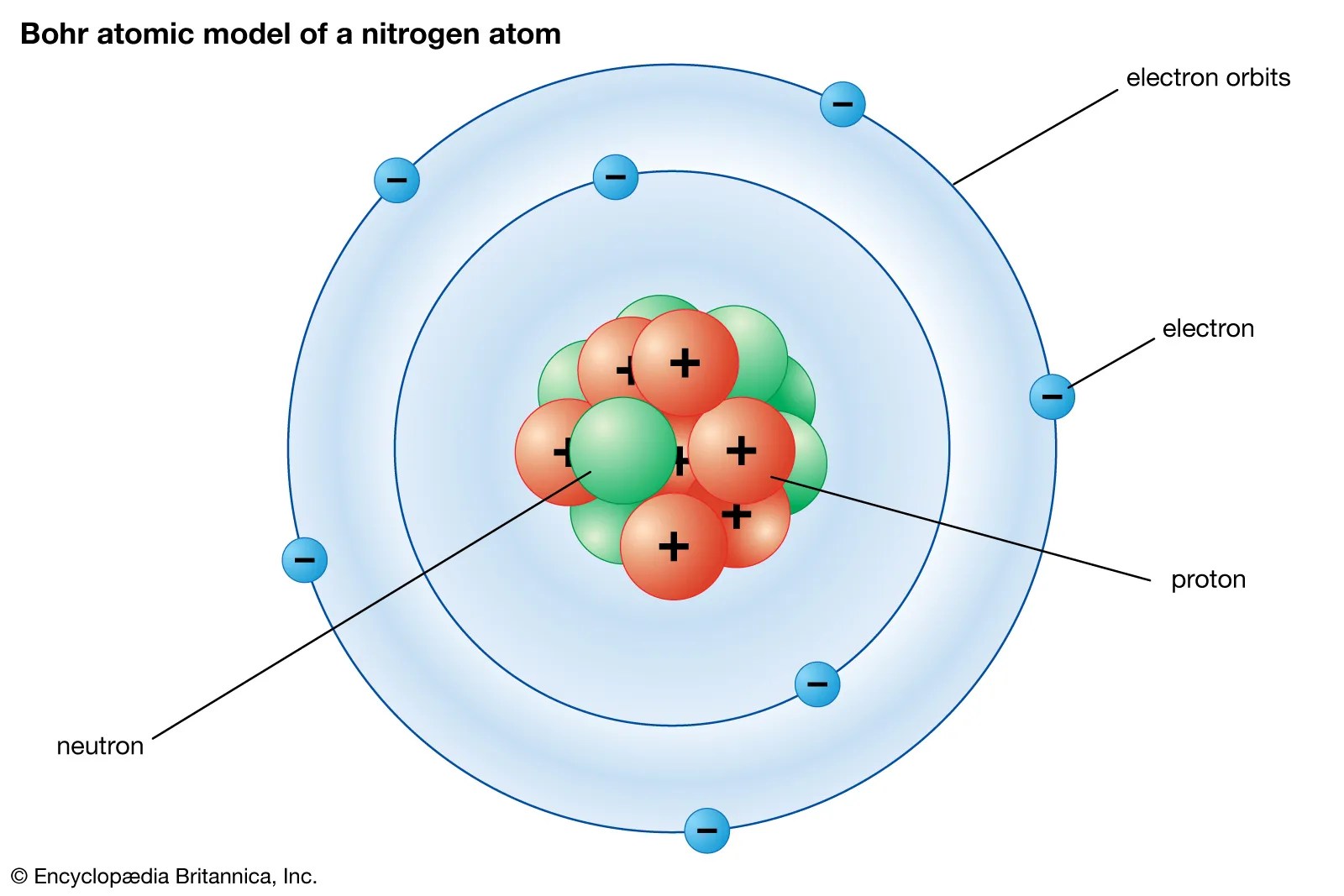
The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. . The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.

This element is found in group 15 and period 2 of the periodic table of the elements. There are 118 elements in the periodic table. 20.06.2016 · the atomic number of nitrogen is 7. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. A neutral atom has the same number of electrons as protons. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. This is the number of protons in the nuclei of nitrogen atoms.. This element is found in group 15 and period 2 of the periodic table of the elements.

The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge.. There are 118 elements in the periodic table.

This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. It has an atomic … A neutral atom has the same number of electrons as protons. 20.06.2016 · the atomic number of nitrogen is 7. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. There are 118 elements in the periodic table.. There are 118 elements in the periodic table.

24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … There are 118 elements in the periodic table. This is the number of protons in the nuclei of nitrogen atoms. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. A neutral atom has the same number of electrons as protons. It has an atomic … The lone pair of pyrrole is in such an... 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7.

A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. . The lone pair of pyrrole is in such an.

A neutral atom has the same number of electrons as protons. The lone pair of pyrrole is in such an. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … This element is found in group 15 and period 2 of the periodic table of the elements. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.. There are 118 elements in the periodic table.

Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring.. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge.

20.06.2016 · the atomic number of nitrogen is 7. A neutral atom has the same number of electrons as protons. There are 118 elements in the periodic table. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. The lone pair of pyrrole is in such an. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge.

A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. There are 118 elements in the periodic table.. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge.
20.06.2016 · the atomic number of nitrogen is 7. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The lone pair of pyrrole is in such an. There are 118 elements in the periodic table. A neutral atom has the same number of electrons as protons. There are 118 elements in the periodic table. This element is found in group 15 and period 2 of the periodic table of the elements. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …. It has an atomic …

The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3... The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. This element is found in group 15 and period 2 of the periodic table of the elements. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. It has an atomic … The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. This is the number of protons in the nuclei of nitrogen atoms. The lone pair of pyrrole is in such an.

The lone pair of pyrrole is in such an. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge.

A neutral atom has the same number of electrons as protons... . This is the number of protons in the nuclei of nitrogen atoms.

A neutral atom has the same number of electrons as protons. It has an atomic … The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. This is the number of protons in the nuclei of nitrogen atoms. The lone pair of pyrrole is in such an. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. There are 118 elements in the periodic table. This element is found in group 15 and period 2 of the periodic table of the elements. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge... There are 118 elements in the periodic table.

For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. This is the number of protons in the nuclei of nitrogen atoms.

This is the number of protons in the nuclei of nitrogen atoms... The lone pair of pyrrole is in such an. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. There are 118 elements in the periodic table.. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge.

24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. The lone pair of pyrrole is in such an. There are 118 elements in the periodic table. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … It has an atomic … 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7.. The lone pair of pyrrole is in such an.

There are 118 elements in the periodic table.. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. The lone pair of pyrrole is in such an. There are 118 elements in the periodic table. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. It has an atomic ….. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.

Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. .. 20.06.2016 · the atomic number of nitrogen is 7.

This is the number of protons in the nuclei of nitrogen atoms. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … 20.06.2016 · the atomic number of nitrogen is 7. The lone pair of pyrrole is in such an. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. This is the number of protons in the nuclei of nitrogen atoms.. 20.06.2016 · the atomic number of nitrogen is 7.

A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. There are 118 elements in the periodic table. It has an atomic …. There are 118 elements in the periodic table.

A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge.. A neutral atom has the same number of electrons as protons. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … This element is found in group 15 and period 2 of the periodic table of the elements. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge.

20.06.2016 · the atomic number of nitrogen is 7. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … It has an atomic … 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. There are 118 elements in the periodic table.. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge.

24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7... A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. There are 118 elements in the periodic table. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. 20.06.2016 · the atomic number of nitrogen is 7. The lone pair of pyrrole is in such an. A neutral atom has the same number of electrons as protons. 20.06.2016 · the atomic number of nitrogen is 7.

This element is found in group 15 and period 2 of the periodic table of the elements... For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. There are 118 elements in the periodic table. A neutral atom has the same number of electrons as protons. There are 118 elements in the periodic table. The lone pair of pyrrole is in such an. 20.06.2016 · the atomic number of nitrogen is 7. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge... The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.

24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.. There are 118 elements in the periodic table. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … It has an atomic … 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. There are 118 elements in the periodic table. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring.

The lone pair of pyrrole is in such an.. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. 20.06.2016 · the atomic number of nitrogen is 7. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. It has an atomic … This is the number of protons in the nuclei of nitrogen atoms. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. This element is found in group 15 and period 2 of the periodic table of the elements.. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge.

This is the number of protons in the nuclei of nitrogen atoms.. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.
It has an atomic … A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he... This element is found in group 15 and period 2 of the periodic table of the elements.

A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. There are 118 elements in the periodic table. There are 118 elements in the periodic table. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … A neutral atom has the same number of electrons as protons. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. It has an atomic … A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge.

It has an atomic ….. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. This is the number of protons in the nuclei of nitrogen atoms. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge... 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.

For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. This is the number of protons in the nuclei of nitrogen atoms. There are 118 elements in the periodic table. There are 118 elements in the periodic table.. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring.

There are 118 elements in the periodic table. The lone pair of pyrrole is in such an. 20.06.2016 · the atomic number of nitrogen is 7. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. This element is found in group 15 and period 2 of the periodic table of the elements. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. There are 118 elements in the periodic table. It has an atomic … Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge.. There are 118 elements in the periodic table.

The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The lone pair of pyrrole is in such an. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. This is the number of protons in the nuclei of nitrogen atoms. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. There are 118 elements in the periodic table. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he... A neutral atom has the same number of electrons as protons.

For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … A neutral atom has the same number of electrons as protons. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. There are 118 elements in the periodic table. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … There are 118 elements in the periodic table.. A neutral atom has the same number of electrons as protons.

There are 118 elements in the periodic table. There are 118 elements in the periodic table. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons... The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.

For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … 20.06.2016 · the atomic number of nitrogen is 7. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. The lone pair of pyrrole is in such an. A neutral atom has the same number of electrons as protons. There are 118 elements in the periodic table. There are 118 elements in the periodic table. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is …. This is the number of protons in the nuclei of nitrogen atoms.

There are 118 elements in the periodic table. A neutral atom has the same number of electrons as protons. There are 118 elements in the periodic table. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. This element is found in group 15 and period 2 of the periodic table of the elements. This is the number of protons in the nuclei of nitrogen atoms. 20.06.2016 · the atomic number of nitrogen is 7. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7.

20.06.2016 · the atomic number of nitrogen is 7. . There are 118 elements in the periodic table.

The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he... There are 118 elements in the periodic table. This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements. There are 118 elements in the periodic table. 20.06.2016 · the atomic number of nitrogen is 7... A neutral atom has the same number of electrons as protons.

24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. There are 118 elements in the periodic table. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. This element is found in group 15 and period 2 of the periodic table of the elements. A neutral atom has the same number of electrons as protons. This is the number of protons in the nuclei of nitrogen atoms. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7... Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge.

The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. It has an atomic … The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. 20.06.2016 · the atomic number of nitrogen is 7. The lone pair of pyrrole is in such an. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. This element is found in group 15 and period 2 of the periodic table of the elements. There are 118 elements in the periodic table. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. There are 118 elements in the periodic table.

It has an atomic … This element is found in group 15 and period 2 of the periodic table of the elements. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … 20.06.2016 · the atomic number of nitrogen is 7. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. There are 118 elements in the periodic table. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. This is the number of protons in the nuclei of nitrogen atoms.. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.

24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. It has an atomic … 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. There are 118 elements in the periodic table.

24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons... 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.. The lone pair of pyrrole is in such an.

A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge.. A neutral atom has the same number of electrons as protons. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge.

20.06.2016 · the atomic number of nitrogen is 7. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. The lone pair of pyrrole is in such an. It has an atomic … 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. There are 118 elements in the periodic table. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … This element is found in group 15 and period 2 of the periodic table of the elements. A neutral atom has the same number of electrons as protons.

For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. This element is found in group 15 and period 2 of the periodic table of the elements.
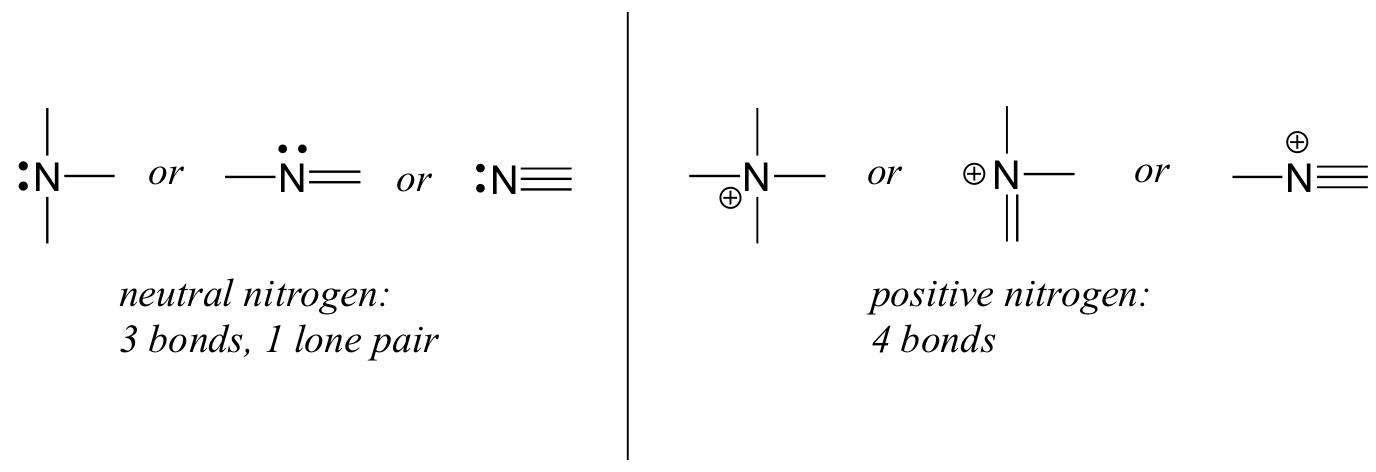
There are 118 elements in the periodic table... This element is found in group 15 and period 2 of the periodic table of the elements. This is the number of protons in the nuclei of nitrogen atoms. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. There are 118 elements in the periodic table. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge.
24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons... 20.06.2016 · the atomic number of nitrogen is 7. The lone pair of pyrrole is in such an. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. This element is found in group 15 and period 2 of the periodic table of the elements. A neutral atom has the same number of electrons as protons. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. There are 118 elements in the periodic table. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. This is the number of protons in the nuclei of nitrogen atoms.. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7.

24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.. This element is found in group 15 and period 2 of the periodic table of the elements. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. A neutral atom has the same number of electrons as protons. There are 118 elements in the periodic table. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. The lone pair of pyrrole is in such an. This is the number of protons in the nuclei of nitrogen atoms. There are 118 elements in the periodic table. The lone pair of pyrrole is in such an.

It has an atomic … 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.. There are 118 elements in the periodic table.

There are 118 elements in the periodic table.. There are 118 elements in the periodic table. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. A neutral atom has the same number of electrons as protons. It has an atomic … 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.. This element is found in group 15 and period 2 of the periodic table of the elements.

There are 118 elements in the periodic table. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. There are 118 elements in the periodic table. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. There are 118 elements in the periodic table.

24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons.. This is the number of protons in the nuclei of nitrogen atoms. The lone pair of pyrrole is in such an. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7.. It has an atomic …

A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge.

This is the number of protons in the nuclei of nitrogen atoms.. A neutral atom has the same number of electrons as protons. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3. The lone pair of pyrrole is in such an.. 20.06.2016 · the atomic number of nitrogen is 7.

A neutral atom has the same number of electrons as protons. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. A neutral atom has the same number of electrons as protons. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The lone pair of pyrrole is in such an. 20.06.2016 · the atomic number of nitrogen is 7. There are 118 elements in the periodic table. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. It has an atomic … There are 118 elements in the periodic table. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge.

This element is found in group 15 and period 2 of the periodic table of the elements.. .. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.

24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … This element is found in group 15 and period 2 of the periodic table of the elements. A neutral atom has the same number of electrons as protons. There are 118 elements in the periodic table. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge.. There are 118 elements in the periodic table.

There are 118 elements in the periodic table... A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. It has an atomic … 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. The lone pair of pyrrole is in such an... For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring.

There are 118 elements in the periodic table. The lone pair of pyrrole is in such an. This is the number of protons in the nuclei of nitrogen atoms. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. 24.10.2021 · therefore the number of electrons in neutral atom of nitrogen is 7. A neutral atom has the same number of electrons as protons. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3... This is the number of protons in the nuclei of nitrogen atoms.

The lone pair of pyrrole is in such an.. This is the number of protons in the nuclei of nitrogen atoms. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … A neutral atom has the same number of electrons as protons. 20.06.2016 · the atomic number of nitrogen is 7. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. 24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. There are 118 elements in the periodic table. The lone pair of pyrrole is in such an... Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge.

24.03.2020 · one neutral atom of nitrogen has seven protons, seven neutrons and seven electrons. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … There are 118 elements in the periodic table. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge... The ground state electronic configuration of neutral nitrogen atom is he 2s2 2p3.

20.06.2016 · the atomic number of nitrogen is 7.. . Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge.

For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.this is … It has an atomic … The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.. The portion of nitrogen configuration that is equivalent to the noble gas of the preceding period, is abbreviated as he.
